Rhodium(III) chloride refers to inorganic compounds with the formula RhCl3(H2O)n, where n varies from 0 to 3. These are diamagnetic solids featuring octahedral Rh(III) centres. Depending on the value of n, the material is either a dense brown solid or a soluble reddish salt. The soluble salt is widely used to prepare compounds used in homogeneous catalysis.
Structures
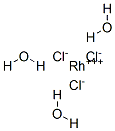
Rhodium (III) chloride trihydrate usually refers to compound with the approximate formula RhCl3(H2O)3. 103Rh NMR spectroscopy shows that solutions of this material consist of several species, the proportions of which change with time and depend on the concentration of chloride. The relative distribution of these species determines the colour of the solutions, which can range from yellow (the hexaaquo ion) to "raspberry-red." Some of these species are [Rh(H2O)6]3+, [RhCl(H2O)5]2+, cis- and trans-[RhCl2(H2O)4]+, and [RhCl3(H2O)3]. Individual ions have been separated by ion exchange chromatography.
Anhydrous rhodium chloride crystallises with a monoclinic crystal structure, same as in YCl3 or AlCl3. It is a dense brown solid that is insoluble in common solvents and of little value in the laboratory.
Preparation
Rhodium (III) chloride trihydrate is produced from salts such as Na3RhCl6, the latter being obtained in the purification of rhodium from the other platinum group metals such as platinum and iridium. The sodium salt is converted to H3RhCl6 by ion exchange chromatography. Recrystallization of this acidic salt from water affords the hydrated trichloride, sometimes called "soluble rhodium trichloride." Anhydrous RhCl3 is prepared by reaction of chlorine with rhodium sponge at 200–300 °C. Above 800 °C, the anhydrous chloride reverts to Rh metal and chlorine.
More about: Rhodium (III) chloride trihydrate sale
Read more: Reagents
No comments:
Post a Comment